FAQ: common questions about the ISO 13485 standard for medical devices

ISO 13485 is the international reference standard for quality management systems in the medical device industry. Although many manufacturers associate it only with a formal requirement for certification, its scope goes much further: it defines how the processes that ensure the safety, efficacy and regulatory compliance of the product throughout its life cycle must be […]
Innovation in biomaterials: how they are validated prior to implantation
Innovation in biomaterials has transformed the design and performance of implantable medical devices. New metal alloys, advanced polymers, technical ceramics and bioactive coatings are enabling the development of implants that are more durable, biocompatible and adapted to today’s clinical requirements. However, before a biomaterial can be used in a surgical implant, it must pass a […]
Testing of femoral stems: the role of the ISO 7206-4 standard
Femoral stems are the main structural component of a hip prosthesis. They must withstand millions of load cycles by reproducing walking, standing, pivoting and other biomechanical stresses. To ensure their safety and durability, the ISO 7206-4 standard establishes the method for testing their fatigue strength. In this article we discuss the importance of this standard, […]
Exoskeletons for rehabilitation: clinical and technical validation.
Exoskeletons for rehabilitation have become a key tool for patients with spinal cord injuries, stroke or musculoskeletal pathologies. To guarantee their therapeutic efficacy and safety, it is essential to carry out a rigorous technical and clinical validation, aligned with medical device regulations. In this article we discuss how exoskeletons are evaluated, what tests are required […]
How coatings affect the durability of surgical implants

Surface coatings are one of the most relevant areas of innovation in modern surgical implants. Biomaterials such as HA (hydroxyapatite), TiN, DLC or ceramic coatings improve osseointegration, reduce friction, minimize wear and extend implant life. But these benefits depend directly on the quality, adhesion and mechanical strength of the coating. In this article we explain […]
What is a four-point bending test and why is it essential?
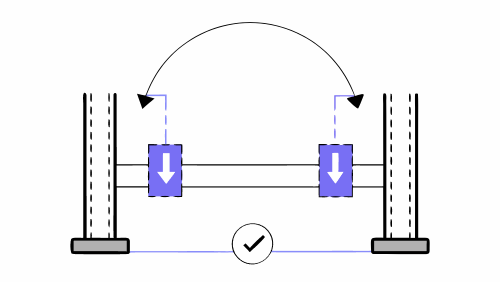
The four-point bending test is one of the most widely used mechanical tests to validate the strength and stiffness of structural components. In the field of medical devices, this test is particularly relevant for evaluating products subjected to repetitive loads and bending stresses, such as osteosynthesis platesinternal fixations, implantable metal components and certain surgical instrumentation […]
Difference between accredited and non-accredited tests on medical devices
Making the right choice between accredited and non-accredited testing is a strategic decision for any medical device manufacturer. It can make the difference between a smooth certification process and one fraught with delays, Notified Body observations or even complete retesting. In this article we look at what distinguishes these two types of testing, in which […]
Hip replacement trials: what does MDR require?
Hip prostheses are one of the most widely used implants in modern orthopedic surgery. Their development combines advanced engineering, high-performance biomaterials and rigorous biomechanical validation.With the entry into force of the European Medical Device Regulation (MDR 2017/745), testing and technical documentation requirements have become more stringent, seeking to ensure greater safety, traceability and clinical evidence. […]
Bone plate assays: from trauma surgery to the laboratory

In traumatologic and maxillofacial surgery, bone plates -also known as osteosynthesis plates– are essential elements for stabilizing fractures, enabling bone healing and restoring skeletal function.Their reliability depends on their mechanical strength, structural rigidity and durability under actual loading conditions. To ensure these factors, manufacturers must perform biomechanical and regulatory tests to validate the performance of […]
MDR 2017/745 explained: key issues for medical device manufacturers
Regulation (EU) 2017/745, known as MDR (Medical Device Regulation), has completely transformed the regulatory landscape for medical devices in Europe. This regulatory framework replaces the old directives (MDD 93/42/EEC and AIMDDD 90/385/EEC), imposing stricter requirements to ensure safety, traceability and transparency throughout the medical device lifecycle. For manufacturers, understanding and correctly applying the MDR is […]

