What is critical load in dental implants?

In dental implantology, one of the most relevant concepts from the biomechanical point of view is the critical load. This term refers to the load level at which an implant can initiate a process of mechanical failure or loss of stability, either due to fatigue, excessive deformation or micro-movements at the bone-implant interface. Understanding and […]
Differences between MDR and MDD for manufacturers
The move from Directive 93/42/EEC (MDD) to Regulation (EU) 2017/745 (MDR) has brought about one of the biggest regulatory changes in the history of the medical device sector in Europe. For manufacturers, the transition has not simply been a regulatory change, but a profound transformation in: In this article we answer the most frequently asked […]
What tests must a dental implant pass before it reaches the market?
The development of a dental implantable medical device does not end when the design is finalized. Before reaching the market, a dental implant must pass a battery of mechanical, biological and regulatory tests that demonstrate its safety, performance and durability in accordance with Regulation (EU) 2017/745 (MDR). For manufacturers, R&D and quality managers, understanding these […]
Why should osteosynthesis plates be validated according to ASTM F382?
Osteosynthesis plates are implantable medical devices designed to stabilize bone fractures and allow proper bone healing. During the healing process, these plates are subjected to repeated loads, bending, torsion and combined stresses, which makes it essential to validate their mechanical behavior before marketing. One of the international reference standards for this validation is ASTM F382, […]
Fatigue testing: what do 5 million cycles mean in clinical practice?
In the development of an implantable medical device, few figures appear as often in technical reports as 5 million cycles. This magnitude is recurrently used in fatigue tests for joint prostheses, dental implants, osteosynthesis components and other products subjected to repeated loads. But what does it really mean to exceed 5 million cycles? How does […]
How osseointegration is measured in dental and orthopedic implants
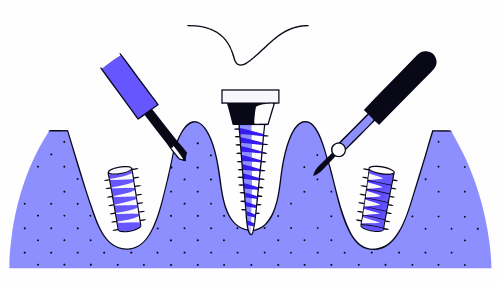
Osseointegration is one of the determining factors for the long-term success of an implantable medical device. In both dental implantology and orthopedic implants, the ability of the implant to integrate stably with the bone determines the load transmission, the durability of the assembly and the clinical safety of the patient. Measuring osseointegration is neither a […]
Trials of tibial trays in knee prostheses
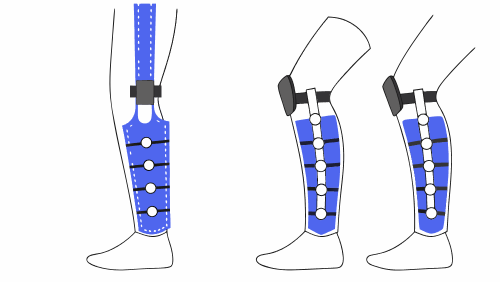
Tibial trays are one of the most critical structural components of a knee prosthesis. Their function is to transmit loads from the femur to the tibia, ensuring stability, alignment and durability over millions of cycles of use. Therefore, the mechanical validation of this component is an essential step in the development of any implantable medical […]
What is a wear test on joint prostheses?
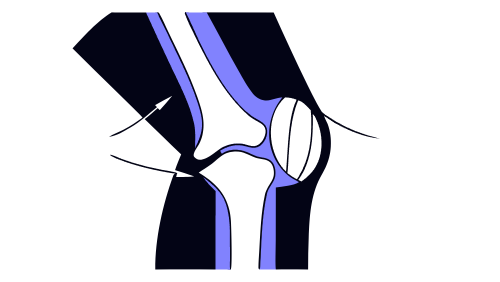
The wear test is one of the most relevant tests in the validation of implantable medical devices intended for joints such as the hip, knee or shoulder. Its objective is to evaluate material loss and particle generation when two joint surfaces interact under repeated loading and movement, simulating real clinical use. Excessive wear can compromise […]
Med-Lab IBV at Expodental 2026: where dental innovation becomes a viable healthcare product

Expodental 2026 will once again bring together the main players in the dental sector in Spain. But beyond the exhibition of new products, this edition takes place at a particularly relevant time for manufacturers and clinics involved in implantology: increased regulatory pressure, accelerated advances in digitalization and a growing demand for customized and clinically robust […]
Exoskeletons: types, applications and certification challenges
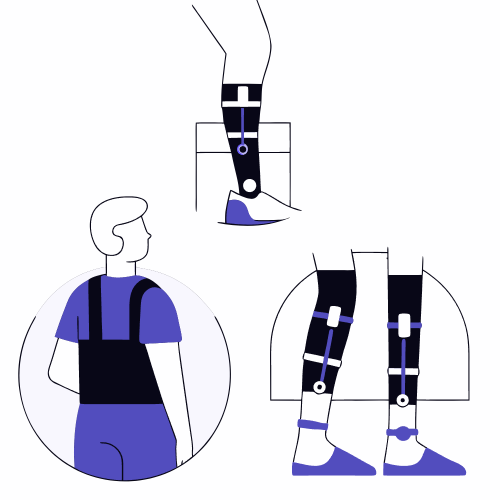
The exoskeletons have established themselves as one of the most innovative technologies in the field of applied biomechanics, with remarkable growth both in clinical rehabilitation and in industrial and healthcare environments. These devices, designed to assist or increase human motor capacity, combine mechanical engineering, electronic control and in-depth knowledge of human movement. However, their increasing […]

