Osseointegration is one of the determining factors for the long-term success of an implantable medical device. In both dental implantology and orthopedic implants, the ability of the implant to integrate stably with the bone determines the load transmission, the durability of the assembly and the clinical safety of the patient.
Measuring osseointegration is neither a simple nor a single process: it requires a combination of mechanical testing, morphological analysis and biomechanical criteria, adapted to the type of implant and its intended use. In this article, we explain how osseointegration is assessed, what methods are used, and how this evidence is integrated into the framework of Regulation (EU) 2017/745 (MDR).
What is osseointegration?
Osseointegration is defined as the direct structural and functional union between the living bone and the implant surface, without intervening fibrous tissue. From a biomechanical point of view, it implies that the implant:
- remains stable under load,
- transmits stresses adequately to the bone,
- does not present excessive micro-movements,
- maintains its fixation over time.
This concept is applicable to both dental implants and orthopedic implants, although evaluation methods may vary according to the anatomical environment and type of loading.
Why is it key to measure osseointegration?
Insufficient osseointegration may cause:
- mobility of the implant,
- loss of primary or secondary stability,
- pain and clinical failure,
- need for surgical reintervention.
From a regulatory standpoint, the MDR requires demonstrating that the medical device fulfills its intended function throughout its lifetime, including justifying that the bone fixation mechanism is adequate and safe.
Methods for assessing osseointegration
The measurement of osseointegration is supported by different complementary approaches, which provide both quantitative and qualitative information.
Mechanical fastening tests
They are essential for assessing the strength of the bone-implant interface. Among the most commonly used are:
- Pull-out tests
Measure the force required to remove the implant from the bone or the simulating substrate. - Push-out tests
Evaluate the resistance to implant displacement in the opposite direction to its insertion. - Torsional tests
Analyze the stability against torsional moments, especially relevant in dental implantology.
These tests make it possible to quantify mechanical stability and to compare designs, surfaces or treatments.
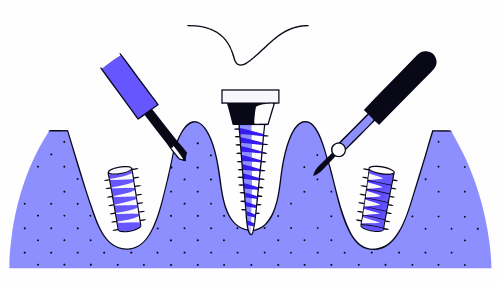
Micro-motion and stability analysis
The presence of excessive micro-movements at the bone-implant interface may prevent proper osseointegration. For this reason, they are used:
- controlled cyclic load tests,
- measurement of relative displacements,
- stiffness analysis of the implant-bone assembly.
These studies allow us to identify whether the design of the medical device favors stable integration under functional load.
Evaluation of implant surfaces and treatments
Osseointegration is closely related to the topography and composition of the implant surface. Therefore, aspects such as:
- surface roughness,
- sandblasting, acid etching or plasma spray treatments,
- bioactive coatings such as hydroxyapatite,
- chemical stability of the surface.
These analyses help to correlate surface characteristics with the biomechanical behavior of the implant.
Differences between dental implantology and orthopedic implantology
Although the principle of osseointegration is common, there are relevant differences:
- in dental implantology, more frequent cyclic loads predominate, but of lesser magnitude,
- in orthopedic implants, the loads are usually higher and more complex (compression, bending, torsion).
This involves adapting test methods and interpretation criteria to each specific application.
Relationship to MDR 2017/745
The MDR requires manufacturers to demonstrate, through technical and scientific evidence, that the medical device:
- maintains adequate fixation to the bone,
- presents no risk of loss of stability,
- has been evaluated with appropriate and traceable methods.
For this reason, osseointegration tests are usually integrated into the technical file and, when safety-critical, are performed in ISO/IEC 17025 accredited laboratories.
How Med-Lab IBV supports osseointegration evaluation
Med-Lab IBV combines capabilities of:
- mechanical fastening tests,
- biomechanical stability analysis,
- evaluation of surfaces and treatments,
- MDR-oriented technical interpretation.
This approach allows medical device manufacturers to properly understand and justify osseointegration, reducing uncertainties and facilitating regulatory validation.