In the development of an implantable medical device, few figures appear as often in technical reports as 5 million cycles. This magnitude is recurrently used in fatigue tests for joint prostheses, dental implants, osteosynthesis components and other products subjected to repeated loads.
But what does it really mean to exceed 5 million cycles? How does this figure relate to the clinical lifetime of the implant? And why is it so relevant for compliance with Regulation (EU) 2017/745 (MDR)? In this article we explain it from a technical and applied perspective.
What is a medical device fatigue test?
A fatigue test evaluates the behavior of a medical device when subjected to repeated cyclic loading, simulating prolonged use under real conditions. Unlike static testing, which applies a maximum point load, fatigue analyzes how the material or structural assembly responds to accumulated damage over time.
These tests allow us to identify:
- crack initiation and propagation,
- progressive deformations,
- failures due to accumulation of damage,
- structural durability limits.
Why are 5 million cycles used?
The figure of 5 million cycles is not arbitrary. It has historically been adopted as a reference value representing several years of clinical use for products subjected to repeated loading, such as occurs in walking or chewing.
In general terms:
- a person can perform between 1 and 2 million walking cycles per year,
- Approximately 5 million cycles is equivalent to 2 to 5 years of functional use, depending on the level of activity.
For this reason, many international standards set this threshold as a minimum to demonstrate that the medical device can withstand a significant period of use without failure.
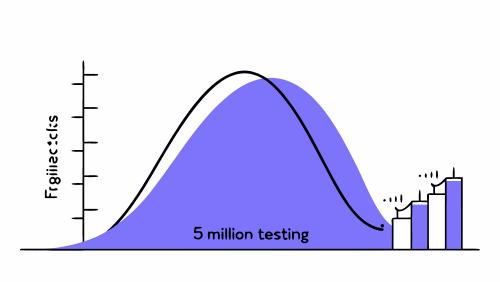
Relationship between test cycles and clinical life
It is important to understand that 5 million cycles does not directly equate to total implant life. In fact, fatigue tests are designed to be conservative:
- the applied loads are usually equal to or higher than the average physiological loads,
- critical alignment or assembly scenarios are tested,
- the aim is to accelerate the emergence of potential failures.
Surpassing 5 million cycles without structural failure indicates that the design has an adequate safety margin, but does not imply that the implant will stop functioning after reaching that number in the clinic.
Medical devices where this criterion is common
The 5 million cycle threshold appears frequently in fatigue tests of:
- femoral stems and hip prosthesis components,
- tibial trays and other knee prosthesis elements,
- osteosynthesis plates subjected to repeated loading,
- dental implants tested under cyclic loading,
- structural components of implantable products.
Each case requires a specific interpretation depending on the intended use and biomechanical environment.
Standards incorporating cyclic fatigue testing
Several ISO and ASTM standards use fatigue tests with millions of cycles as validation criteria, among them:
- ISO 7206-4 (femoral stem fatigue),
- ISO 14801 (fatigue in dental implants),
- ASTM F382 (fatigue in bone plates),
- specific standards for joint prostheses and structural components.
The number of cycles, load level and test setup vary depending on the standard and the medical device being tested.
What happens if the implant does not exceed 5 million cycles?
Failure to exceed this threshold may indicate:
- unanticipated stress concentrations,
- inadequate material selection or surface treatment,
- geometric design problems,
- test conditions too demanding for the intended use.
In these cases, fatigue testing allows for design feedback, geometry optimization and risk reduction before moving towards certification.
Regulatory interpretation under the MDR
The MDR requires proof that the medical device:
- maintains its performance throughout its expected service life,
- does not present unacceptable risks due to mechanical failure,
- has solid and traceable technical evidence.
Fatigue tests with millions of cycles are a key part of the technical dossier, especially for implantable products. When these tests are performed in ISO/IEC 17025 accredited laboratories, their acceptance by notified bodies is significantly higher.
Med-Lab IBV’s approach to fatigue testing
Med-Lab IBV designs and executes fatigue tests adapted to the real use of the medical device, considering:
- representative load configurations,
- stress levels in accordance with clinical use,
- realistic mounting conditions,
- biomechanical interpretation of the results.
This approach allows manufacturers to understand what exceeding 5 million cycles really means for their particular product and how to properly justify it to the regulatory assessment.