Tibial trays are one of the most critical structural components of a knee prosthesis. Their function is to transmit loads from the femur to the tibia, ensuring stability, alignment and durability over millions of cycles of use. Therefore, the mechanical validation of this component is an essential step in the development of any implantable medical device for knee arthroplasty.
In this article we explain how tibial trays are tested, what tests are key to demonstrating their mechanical behavior, and what Regulation (EU) 2017/745 (MDR) requires of manufacturers to ensure safety and clinical performance.
Why are tibial tray assays critical?
During walking, going up and down stairs or changes of direction, the knee prosthesis is subjected to:
- high compression loads,
- bending and torsional stresses,
- repeated eccentric loads,
- micro-movements at the bone-implant interface,
- continuous contact with the polyethylene insert.
The tibial tray must maintain its structural integrity and stability in the face of all these scenarios, avoiding deformations, cracks or premature failures that compromise patient safety.
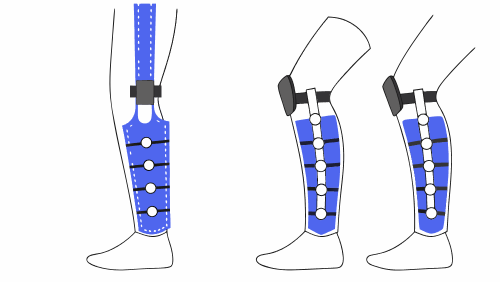
Components and configurations of tibial trays
The tibial trays can have different design configurations, which directly conditions the required tests:
- Monobloc metal trays
- Modular trays with polyethylene insert
- Cemented and uncemented designs
- Smooth or osseointegration treated surfaces
Each variant introduces differences in load distribution and potential failure mechanisms, which must be evaluated experimentally.
Typical mechanical tests on tibial trays
The mechanical validation of this medical device is based on a set of standardized tests and specific tests adapted to the design.
Static structural strength tests
They allow to evaluate the capacity of the tibial tray to withstand maximum loads without permanent deformation or structural failure. They are particularly relevant to justify safety margins against extreme loads.
Fatigue tests
They simulate the behavior of the component under millions of load cycles, reproducing prolonged use in real clinical conditions. These tests make it possible to detect:
- crack initiation,
- accumulation of fatigue damage,
- progressive deformations,
- failures associated with voltage concentrations.
Bending and eccentric load tests
They analyze the behavior of the tibial tray when loads are not applied in a perfectly centered manner, a common situation in patients with non-ideal alignments or during complex movements.
Interface tests with the tibial insert
They evaluate the stability of the tray-insert assembly, the distribution of contact pressures and the risk of micro-movements that could accelerate wear or generate particles.
Applicable reference standards
Tibial tray testing is usually based on recognized international standards, adapted to the specific implant design, including:
- ISO 14879 (when applicable to tibial components),
- ISO 7206 (fatigue criteria applicable by structural analogy),
- specific ASTM standards for joint replacement components,
- custom protocols when the design does not completely fit into a standard norm.
The use of well-defined test methods is key for results to be accepted by notified bodies.
Relationship to MDR 2017/745
The MDR requires demonstrating that implantable medical devices:
- are mechanically safe,
- maintain their performance throughout their expected service life,
- do not introduce unacceptable risks for the patient,
- have traceable and reproducible technical evidence.
In the case of tibial trays, mechanical tests are a central part of the technical dossier, as they allow the durability, structural stability and clinical suitability of the component to be justified.
How Med-Lab IBV supports validation of tibial trays
Med-Lab IBV has advanced capabilities for the evaluation of knee prosthesis tibial trays, including:
- static mechanical and fatigue tests,
- load configurations adapted to the actual implant design,
- analysis of results from a biomechanical and regulatory approach,
- issuance of traceable technical reports aligned with MDR.
This approach allows manufacturers to reduce uncertainties, optimize design prior to certification and minimize retesting risks.